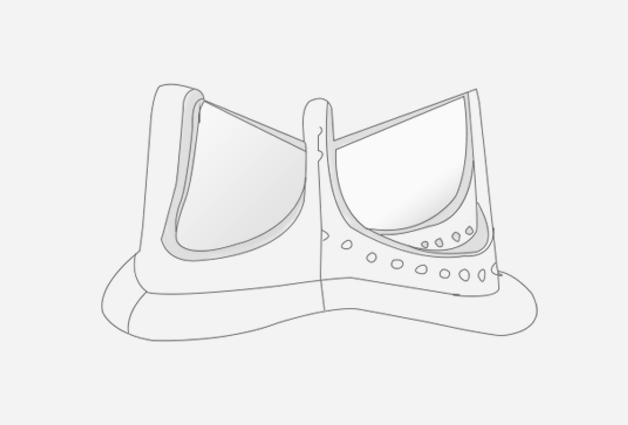
Product introduction:
Indications: aortic stenosis and aortic valve incompetence.
China registration information::
Name of medical device: Bovine Pericardium Heart Valve ProsthesisProduction Enterprise: Shanghai Cingular Biotechnology Co., Ltd.
Medical device registration certificate No.: 国械注准20203130638
Product technical requirements No.: 国械注准20203130638
Specifications and Models:
19A、21A、23A、25A、27A